Understanding Opioids, And Visual Snow Research
Studies have previously mentioned opioids as a potential cause of visual snow but what are opioids really? How could opioid effects contribute to visual snow in theory, and why have they been looked over in practice?
Introduction To Opioids
Most of us when we think of opioids probably only think of drugs, used medically (or abused illicitly) for pain relief. This is because until very recently opioids were understood entirely through the lens of opioid drugs. The extraction of opium for drug use far precedes ideas about why and how these substances actually work – that there are opioid receptors and natural opioids produced within the body.
- The first recorded use of opium in 3400BC.
- Morphine extracted in 1803.
- In 1898 heroin was marketed as a “non-addictive” alternative to morphine.
- Opioid receptors discovered in 1972 (5372 years later).
- Endogenous opioids discovered in 1978.
- In the 1990’s medical use of opioids soared as did opioid abuse.
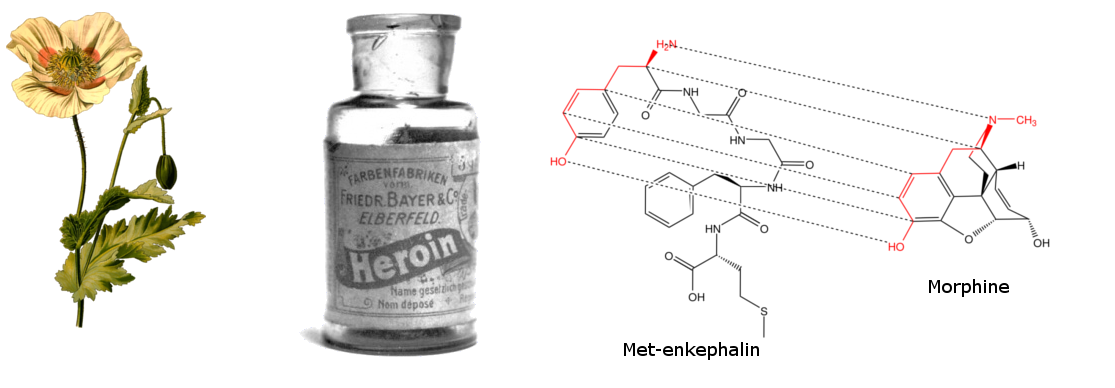
Endogenous And Exogenous Opioids
The natural opioids produced within the body – endogenous opioid peptides, are small molecules produced within the central nervous system (CNS) and various glands. They were discovered to function as neuromodulators and hormones:
- As neuromodulators they modulate the actions of other neurotransmitters
- As hormones they directly regulate the activity of cells and organs
All opioids work by binding onto opioid receptors, which are found on cell surfaces: tissue, neurons, but also inflammatory cells and immune cells. By activating these receptors they elicit chemical changes. Three main types of opioid receptors are commonly identified: mu, kappa, and delta. Different opioids bear different affinities for these receptors – which, because they are found in different areas of the body also have different effects:
- Emotions
- Neuroendocrine physiology ( hormonal interactions between the nervous system and endocrine system) e.g. Respiration, blood pressure, thermoregulation, and gastrointestinal motility.
- Auditory processing
- Cognitive functioning
- Immune responses
- Stress responses
- Cardiovascular
- Motor activity
- Seizure threshold
- Water and food intake
To summarise: internally produced “endogenous” opioids are naturally released to help us react appropriately to stimuli (such as pain) and maintain homeostasis (bodily balance). Externally introduced “exogenous” opioids have the potential to disrupt this system.
Opioid Effects On Vision Versus Visual Snow Research
Opioid receptors are found throughout the visual system, and opioids can also cause significant changes to vision. Early studies found that opioids can cause decreased visual sensitivity and abnormal visual discrimination.
Cortical Hyperexcitability
In visual snow research, initial connections were made with hallucinogen perception persisting disorder (HPPD) and migraine, although it was later found to be distinct from both of these conditions. Through this line of enquiry it however followed that visual snow could be the result of a cortical hyperexcitability Most simply viewed as too much excitation, or not enough inhibition in the brain.
The balance of inhibition to excitation is important for proper visual function and behaviour. Gamma-Aminobutyric acid (GABA) is considered to be the main inhibitory neurotransmitter, whereas glutamate is considered to be the main excitatory neurotransmitter.
Morphine, Acting Via Mu Receptors Can Shift The Balance Towards Excitation:
- Indirectly through neurotransmitter inhibition: morphine inhibits GABA release ( GABA is specifically responsible for stimulus selectivity and the inhibition of visual neurons).
- Directly: morphine alters glutamate synapse morphology to increase excitatory output.
The Consequences From Chronic Morphine Use To Key Visual Areas:
- Lower-signal to noise ratios
- Raised visually evoked responses
- Raised spontaneous activity (neural oscillation)
- Decreased orientation and direction sensitivity (perception of form and motion)
(Lateral Geniculate Nucleus, LGN, Primary Visual Cortex, V1)
-
- Decreased contrast sensitivity (V1)
Magnocellular-Parvocellular-Koniocellular Imbalance
An imbalance of the magnocellular-parvocellular-koniocellular pathways has also been suggested to contribute to symptoms of visual snow. Imbalances, deficits and timing delays in these pathways are debated for their exact role and nature in many neurological conditions including Parkinson’s, schizophrenia, migraine, dyslexia, autism, and Alzheimer’s.
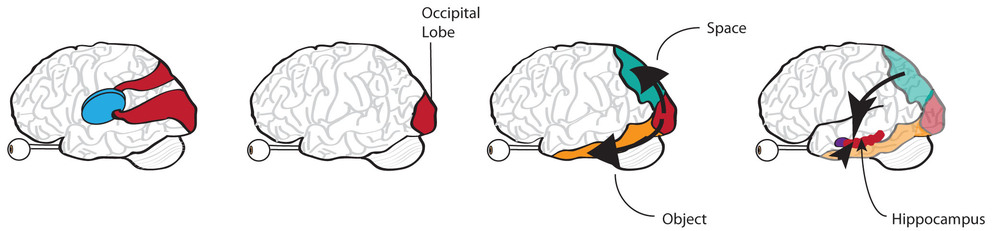
Visual System
In the retina there are several parallel channels formed up of different cells that extract different attributes from the visual scene (there is also a suggestion that visual snow could originate in some of these cells).
The different retinal cells provide the input for koniocellular, parvocellular and magnocellular cells in the LGN, which process different visual information. Considering their functions it is understandable how issues may also contribute in visual snow to pattern glare, contrast sensitivity, poor depth perception, afterimages etc.
Parvocellular (P) – Shape, colour (red-green), high spatial contrast sensitivity, low temporal contrast sensitivity.
Magnocellular (M) – Motion, luminance, high temporal contrast sensitivity, low spatial contrast sensitivity.
Koniocellular (K) – Colour (yellow-blue), exhibit slow rhythmic activity
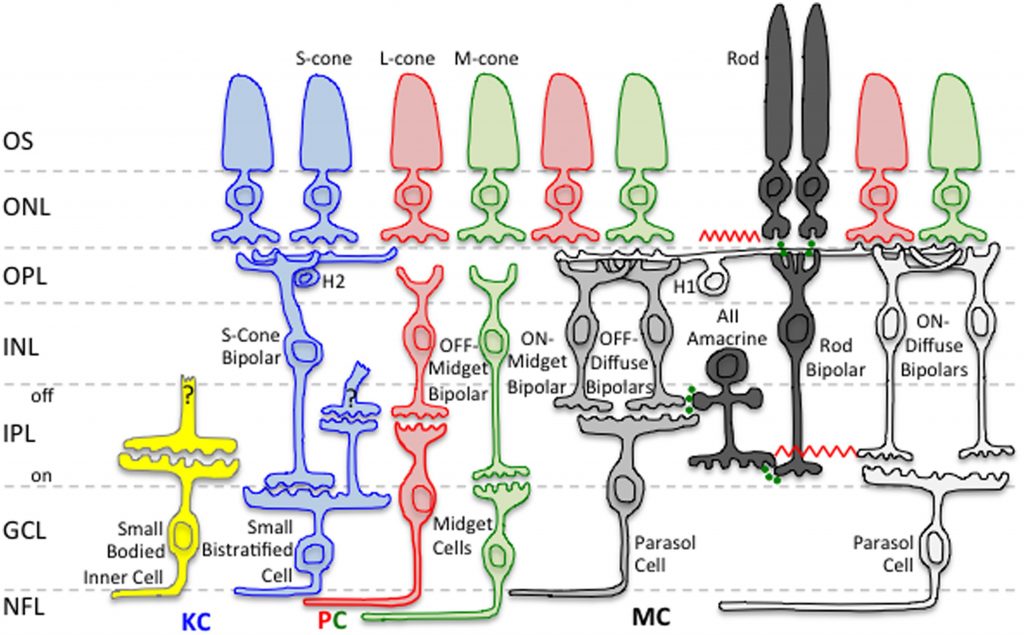
From there that information is sent to the V1 and other parts of the visual cortex, where the magnocellular and parvocellular cell pathways can be said to terminate.
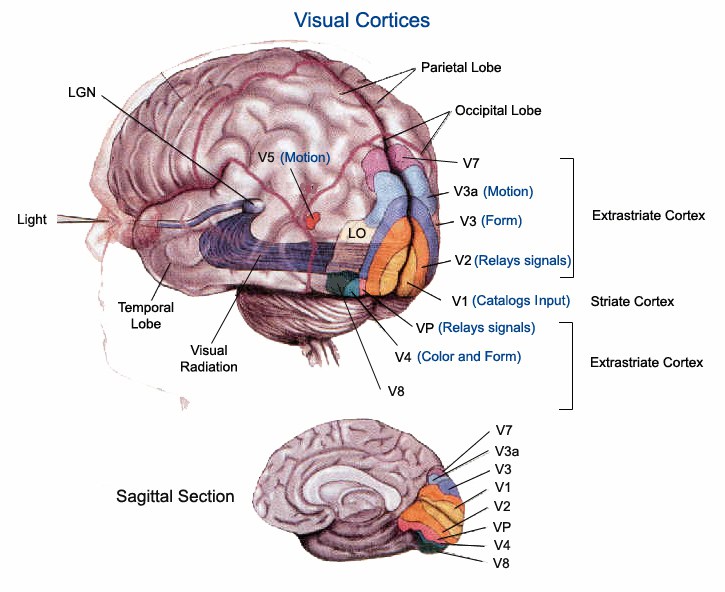
The visual cortex does however send information further, to cognitive areas of the brain. This is believed to be through two pathways called the dorsal and ventral streams. The main input for the dorsal stream comes from the M cells, the main input for the ventral stream comes equally from the P and M cells. K cells, project beyond the visual cortex and their slow cortical frequencies have been shown to modulate high frequency oscillations. P and M cells are linked with high cortical frequencies, and it follows that K cells therefore may modulate the input of P and M cells past the LGN.
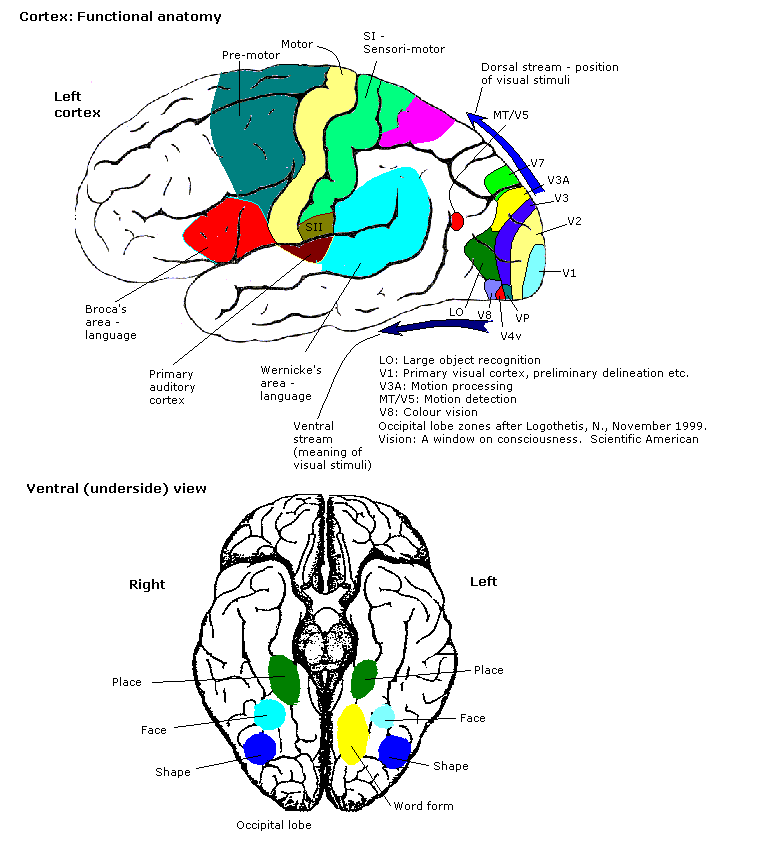
The dorsal and ventral streams integrate increasingly complex additional non-visual information, before they eventually combine at the hippocampus.
Thalamocortical Dysrhythmia
Specifically an imbalance between the M-P-K pathways in visual snow was suggested to underlie a thalamocortical dysrhythmia (TCD) because it was found that yellow tinted lenses can temporarily improve symptoms, and visual stimuli such as yellow tinted lenses that selectively activate k cells may cancel low frequency brain rhythms which contribute to this dysrhythmia (and which as mentioned above may modulate the P and M input).
A TCD is suggested to arise from the excess inhibition of thalamic neurons and inhibitory asymmetry in cortical areas. The thalamic inhibition increases low frequency rhythmicity which can in turn cause a cascade of disruptive events, and excitation in neighbouring cortical parts of the brain.
Mu opioid agonists like morphine can not only affect early parts of the M-P-K pathways (V1, LGN) but also inhibit thalamic neurons and influence thalamocortical activity as per a TCD. After all, migraine and chronic pain from which these theories stem are fundamentally involved with pain and therefore mu opioid receptors.
Additional points to consider
-
- Metabolic requirements (e.g. differences between magnocellular and parvocellular cells). More activity requires more energy, size would also physically impact on a cell’s sensitivity to noise – koniocellular cells are the smallest.
- A fairly recent study also interestingly found that low frequency activity originating from the hippocampus mediates brain wide connectivity and visual processing in both cortical and sub cortical areas. Mu opioids also alter hippocampal oscillatory activity (Gamma, Theta).
The Bottom Line
Tinted lenses only modulate an imbalance between the M-P-K pathways which may or may not be caused by a thalamocortical dysrhythmia as suggested. In any case something would have to cause a dysrhythmia and/or hyperexcitability, and to this end visual snow research has not yet advanced very far. Little is known about many of the important aspects upon which these possibilities hinge.
The Perceived Issues With Opioids
Despite a theoretical fit there are two clear perceived issues
1) The use of opioid drugs appears uncommon among visual snow sufferers.
2) Time gaps between opioid drug use and the onset of visual snow appear too long to link the two.
So if opioids were to be involved with visual snow in practice there would:
a) Have to be a continuous opioid trigger.
b) The mechanism of action would potentially not be so direct.
But are all opioids uncommon among visual snow sufferers? Perhaps the real issue is a historic one – that opioids are being viewed only through an opioid drug lens…


Thank you so much for this information! My daughter is 12 and just got this rare disease Oct 2017, it’s getting worse week by week so anything helps. We are starting this dairy free diet plan to see if it gives her benefit. Thank you again !